 
It basically takes the voltage of the anode and the cathode. By convention in standard cell notation, the anode is written on the left and the cathode is written on the right. Identify the oxidation and reduction reactions. Give the direction of the current in the external circuit. Write the overall equation for the chemical reaction. Galvanic Cells Galvanic cells traditionally are used as sources of DC electrical power. These are half equations for some reactions at the anode: 2Cl- Cl2 + 2e- 2O2- O2 + 4e- Oxidation and Reduction. Give the anode and cathode half-reactions. Both galvanic and electrolytic cells will consist of two electrodes (an anode and a cathode), which can be made of the same or different metals, and an electrolyte in which the two electrodes are immersed. If the anode has a more negative electrode potential than the cathode, electrons will flow. This calculator calculates the value of the electromotive force of any galvanic (volcanic) cell. Anode reactions Negatively charged ions lose electrons at the anode. from decomposition of both the cathode and the anode with electrolyte. The half equations can be combined to form the full equation. The results of this experiment can lead to a discussion about electroplating. The reaction is the reverse of the cathode reaction. If copper is used for the electrodes, the copper anode dissolves. With carbon (graphite) electrodes, the oxygen usually reacts with the anode to form CO 2. The cathode has a contribution based on its ability to gain electeons, its 'reduction potential'. This web-based calculator estimates heats of reaction associated with thermal. Anode reaction: 2H 2 O (l) O 2 (g) + 4H + (aq) + 4e. 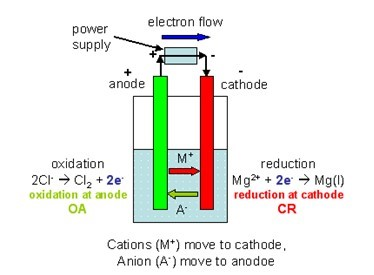 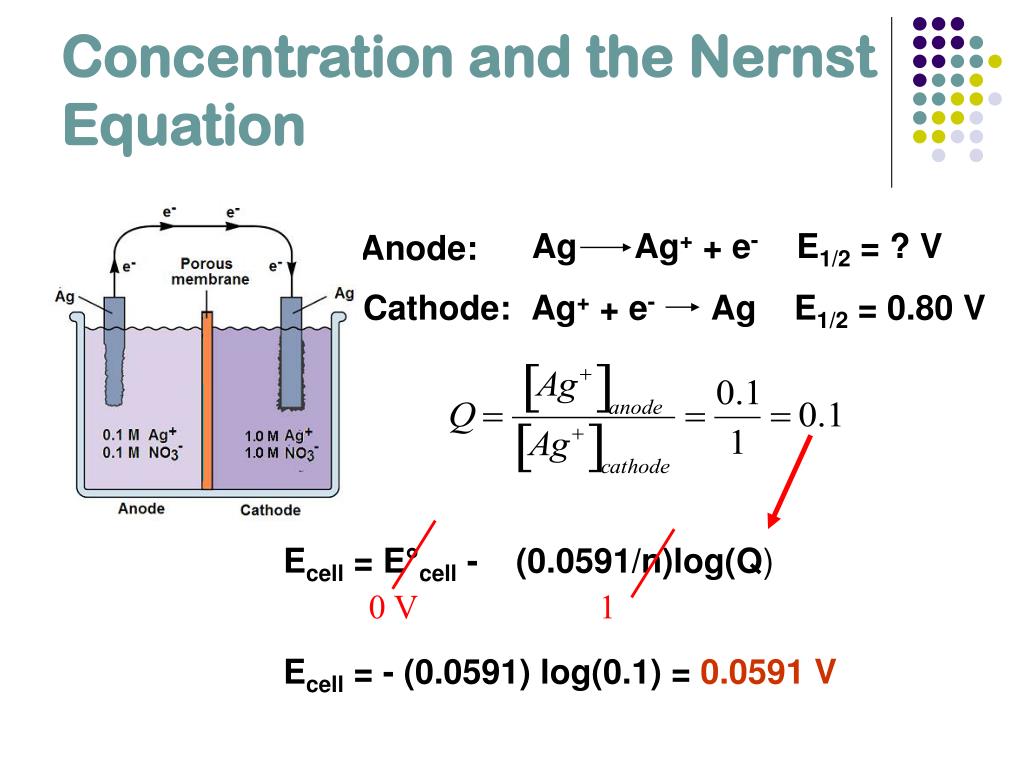
In this problem I'm asked to calculate the electrode potential of the magnesium electrode ($E_$ is a product (not a reactant), therefore following what I studied, its concentration should be in the numerator.\): The Standard Hydrogen Electrode. The cell potential (often called the electromotive force or emf) has a contribution from the anode which is a measure of its ability to lose electrons - it will be called its 'oxidation potential'. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
